The length of the bond increases in the following order- triple bond < double bond < single bond. The short bonds are formed because of an extreme force of attraction. The higher the bond orders are, the stronger bonds that are further accompanied by the robust forces of attraction that hold the atoms together and close to one another. The bond length is generally evaluated by the bond order or the total number of bonded electrons.įor the covalent bonds, the length of the bond is inversely proportional to the order bond. The bond length can be described as the total distance between the centres of 2 covalently bonded atoms. The bond parameters play a significant role in offering insights into the chemical compound stability and chemical bonds strength that holds its atoms together. The Pi bond has nearly no role in determining the molecule’s shape.īond parameters can be defined as the covalent bonds characterised based on multiple bond parameters, including bond length, bond angle, bond order, and bond energy, popularly known as the Bond Enthalpy. The sigma bond has an impact on the molecular shape. The Pi bond needs the support of the sigma bond to exist. The sigma bonds have high bond energies and are comparatively stronger. The overlapping of the orbitals should be unhybridised. The overlapping of orbitals can either be hybrid or pure. Here are the key differences between the Sigma and Pi bonds – Difference between the Sigma and Pi bonds Note that the sigma and pi bonds combination is always better and stronger as compared to the single sigma bond. 
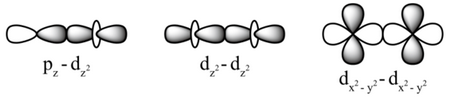
However, a typical triple bond leads to the formation of one σ bond and two π bonds. Mostly, a double bond holds one pi bond and one sigma bond. Compared to sigma bonds, the Pi bonds are generally weaker because they hold a lower degree of overlapping. When the Pi bond forms, the atomic orbitals axes are parallel to one another however, the overlapping is directly perpendicular along with the internuclear axis. These are formed through the sidewise positive or the same phase overlapping the atomic orbitals with the direction perpendicular to the internuclear axis. However, the Pi bond is generated with the lateral overlapping of these orbitals. If there are two p orbitals, head-to-head overlapping gives rise to a sigma bond. 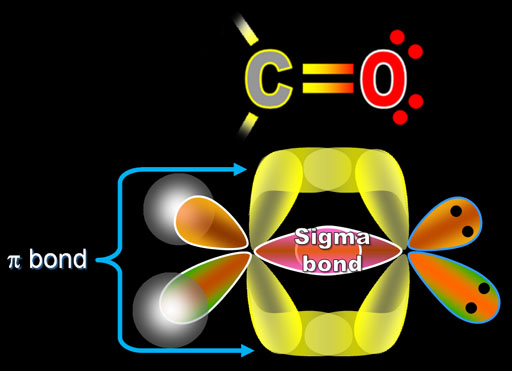
This kind of overlapping mostly occurs in Cl2 In the p-p overlapping, one half-filled p orbital from all-atoms taking part undergoes head-on overlapping with the internuclear axis. This kind of overlapping mostly occurs in ammonia. In the s-p overlapping, the half-filled s orbital is overlapped by the one half-filled p orbital with the internuclear axis that forms a covalent bond. This kind of overlap mostly occurs in H2 s-p Overlapping When two s orbitals overlap, it leads to the sigma bond formation. Before the “s” orbital overlaps with another, it should be half-filled. In the S-S overlapping, one “s” orbital from every atom taking part goes for the head-on overlapping with the internuclear axis. These are formed through several combinations of the atomic orbital. It is mostly seen that all the single bonds are only the sigma bonds. The participating electrons in the sigma bond are popularly known as the σ electrons. Compared to other covalent bonds, the sigma bonds are the strongest as they directly overlap the participating orbitals. The sigma bond is denoted by (σ), a covalent bond primarily formed by head-on positive, also known as the same phase overlap of the atomic orbital and internuclear axis. Without any further ado, let’s get started! What is a Sigma bond? Today, in this article, we will discuss the Sigma and Pi bonds in detail along with other related concepts. Several bond parameters, including the bond length, bond angle, and bond enthalpy, are highly dependent on how atomic orbitals overlap. Note, the sigma bond is denoted by (σ) however, the pi bond is denoted by (π). The sigma bonds are formed by the head-to-head overlapping of the atomic orbitals, whereas the pi bonds are formed by the lateral overlap of two atomic orbitals.īoth Sigma and Pi are Greek words. This overlapping ultimately forms the covalent bonds. The Sigma and Pi bonds are distinguished by the overlapping of atomic orbitals.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
